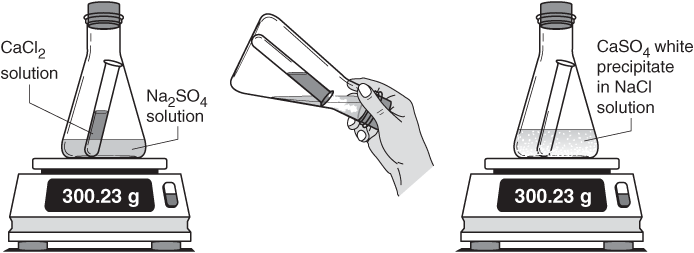
According to the law of conservation of matter, matter is neither created nor destroyed, so we must have the same number and type of atoms after the chemical change as were present before the chemical change.īefore looking at explicit examples of the law of conservation of matter, we need to examine the method chemists use to represent chemical changes. 
Both the initial and final substances are composed of atoms because all matter is composed of atoms. 
What does this mean for chemistry? In any chemical change, one or more initial substances change into a different substance or substances. A concise way of expressing this law is to say that the amount of matter in a system is conserved. It states that in any given system that is closed to the transfer of matter (in and out), the amount of matter in the system stays constant. One scientific law that provides the foundation for understanding in chemistry is the law of conservation of matter. In classical physics, laws of this type govern energy, momentum, angular momentum, mass, and electric charge. Scientific laws form the core of scientific knowledge. conservation law, also called law of conservation, in physics, a principle that states that a certain physical property (i.e., a measurable quantity) does not change in the course of time within an isolated physical system. Laws are therefore considered the highest form of scientific knowledge and are generally thought to be inviolable. Before being accepted, a law must be verified many times under many conditions. In science, a law is a general statement that explains a large number of observations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
